Simple model of intestine developed from stem cells

By Amy Ratner, Medical and Science News Analyst
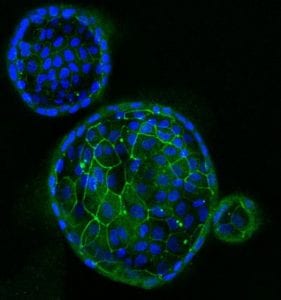
A three-dimensional “mini gut” grown from stem cells in tissue obtained from the biopsies of patients may help researchers unlock some of the mysteries of celiac disease.
Scientists at the Mucosal Immunology and Biology Research Center at Massachusetts General Hospital in Boston developed the intestinal organoids, described as miniature, simplified versions of the intestine produced outside the body. The tissue came from biopsies of those who have celiac disease and those who do not.
Intestinal stem cells from the tissue divide and naturally organize themselves as if they were on the surface of a ball, creating a structure similar to the inside space of an intestine, said Stefania Senger, PhD, co-senior author of a study in Scientific Reports that details the research.
This “very simple model” has enabled researchers to study how multiple factors modify the function of the intestinal cells in regard to gluten, Senger said.
Role of bacteria
“Our intestinal bacterial flora naturally releases a lot of factors that can be beneficial for our health, and they help produce essential vitamins,” she noted. “While we understand the value and function of vitamins, there are many other factors secreted by these bacteria whose function we don’t understand.”
Additionally, researchers don’t know if the secretions are always beneficial or if the effect they have on a person changes based on genetic makeup.
However, scientists have known for a long time that gluten causes inflammation in the gut of celiac disease patients. Observation of the celiac disease mini gut has shown that some secretions from bacteria can reduce inflammation in response to gluten.
“This result is interesting and encouraging,” Senger said. But she noted that the gut is exposed to millions of bioproducts that act on cells at the same time. “We do not know whether the overall effect is the sum of each of them or [whether] some can overrule others,” she said.
The mini-gut might fill a void caused by lack of an animal model for celiac disease that would allow researchers to study the complexities of reactions to gluten. Although animal models have been investigated, there is currently none that would spontaneously develop celiac disease.
| Intestinal epithelium: a single layer of cells that lines the gastrointestinal tract. It is the most rapidly renewing tissue in the body. |
The miniature version of the intestine faithfully mimics the function of the intestinal epithelium of a person with celiac disease, according to Senger. Ongoing work includes improving the model by adding other cell types that are important to the reaction to gluten. Organoids that resemble the brain, kidney, lungs, stomach, liver and more have been developed and many more are being worked on, according to the Harvard Stem Cell Institute.
“We aim to develop a faithful in vitro model to accelerate our understanding of the disease and the identification of therapies for its prevention and cure,” Senger said.
In the study, researchers used RNA sequencing to identify 472 genes regulated differently in gut models of those who have celiac disease compared to those who don’t. Included were genes related to maintaining the gut barrier, stem cell regulation and the innate immune response.
Fellow researchers from the Center for Celiac Disease Research and Treatment at Mass General also recently did a study related to RNA and celiac disease. They looked at at a broader variety of cells in patients’ biopsies. They found 945 differences in genes between those who have celiac disease and those who don’t.
Major shift in celiac disease study
Senger’s study also found that bioproducts from gut microorganisms can be used to modify the intestinal epithelial response to gluten. This discovery could lead to future treatment strategies, according to the study authors.
“The results confirm our hypothesis that genes and exposure to gluten are necessary, but not sufficient since changes in both the composition and function of the gut microbiome are also needed to switch [from having] the genetic predisposition to clinical outcome,” said Alessio Fasano, MD, director of the immunology and biology research center and a co-senior author of the study.
The study represents a “major shift in the study of celiac disease,” Senger said. She noted that with more funding it could lead to development of drug screening to quickly identify new treatments. Also, expansion of the organoid repository could result in development of more complex gut models and an increase in personalized treatment.
You can read the study here.