3 pharmaceutical companies announce changes related to celiac disease drugs under study

By Amy Ratner, Medical and Science News Analyst
Three big players in research into new treatments for celiac disease made significant financial moves this fall.
Most recently, Takeda Pharmaceuticals obtained the exclusive global license of an immunotherapy developed by Cour Pharmaceuticals that targets only the cells that cause a reaction to gluten. Takeda will make $420 million in future payments and royalties on sales of any commercialized products resulting from the license.
In early October, Innovate Biopharmaceuticals, the drug company that is the first to launch a Phase 3 clinical trial of a treatment for celiac disease, announced it will merge with RRD Pharma, Ltd., a start-up company focused gastrointestinal disorders.
The company will have a new name, 9 Meters Biopharma, Inc., which was chosen because the length of the intestinal tract is about nine meters long.
“People living with gastrointestinal diseases, such as celiac disease, are waiting for treatment options,” Nissim Darvish, MD, a member of RDD board of directors, said in a press release. “The proposed merger will create a powerful platform company with a pipeline of late development-stage therapeutic candidates already demonstrating proof of concept.”
In September, GlaxoSmithKlein (GSK) acquired Sitari Pharmaceuticals, which is developing transglutaminase 2 (TG2) inhibitors that have the potential to suppress the autoimmune response triggered by gluten in those who have celiac disease. The enzymatic activity of TG2 is thought to be responsible for starting the inflammatory cascade in response to gluten.
“It is very encouraging to see multiple celiac disease drug development programs meeting their study end points in the past few months and moving on to the next step in the drug development pipeline,” said R. Bennett Muskin, a member of the Beyond Celiac board of directors and managing director at Sagebrook Partners, LLC. “It is also very encouraging to see these programs attract big pharma partners upon achieving development milestones.” He noted that transactions like the ones made recently in celiac disease drug development help validate the programs’ therapeutic value proposition.
| Value proposition: promise by a company to a customer or market segment that is an easy-to-understand reason why a customer should buy a product or service from that particular business. In drug development, value has to be established for patients, providers and payers. |
“Equally important, these transactions provide the resources needed to move the programs through the pipeline towards a new drug application as quickly and efficiently as possible,” Muskin said. “Our hope is that these recent successes will help focus attention on celiac disease as a serious autoimmune disease and an urgent unmet medical need and lead to additional research and funding.”
Takeda
Takeda, an international pharmaceutical company with global headquarters in Japan and US headquarters in Massachusetts, plans to begin a larger study to determine the dose range of the Cour nanoparticle drug, called, CNP-101/Tak-101, in treating celiac disease. Takeda’s licensing decision was based on the recent success of a small, early study testing whether the drug was safe and effective when study participants ate gluten while receiving treatment, according to the company.
Meanwhile, Cour will continue to work on treatments for a variety of other autoimmune diseases in its drug pipeline. “Our collaboration with Cour has shown, for the first time, that it is possible to induce specific immune tolerance to a foreign antigen in autoimmune diseases such as celiac disease,” said Asit Parikh, MD, head of Takeda’s gastroenterology therapeutic unit.
9 Meters focuses on GI diseases
9 Meters Biopharma will focus on developing urgently needed treatments for specialty gastrointestinal diseases including celiac disease and rare and orphan patient populations with gastrointestinal diseases.
In the United States, a rare disease is as a condition that affects fewer than 200,000 people. Rare diseases became known as orphan diseases because drug companies were not interested in developing treatments for them. The Orphan Drug Act created financial incentives to encourage companies to develop new drugs for rare diseases. Celiac disease, once considered rare, is now thought to affect 3 million people in the United States, though many remain undiagnosed.
“With a diversified pipeline of novel compounds, active clinical studies – including the first-ever drug to enter a Phase 3 registration trial in celiac disease – 9 Meters Biopharma expects to deliver multiple value-creating milestones over the next 24 months,” said John Temperato, the current CEO of RDD, who will become the CEO of the new company.
Several clinical trials involving more than 800 patients and a phase 2b trial with 342 patients, have shown that larazotide is safe and effective for celiac disease patients in a real world setting, according to Innovate.
Since the first scientific discoveries that led to the development of larazotide acetate as a potential treatment for celiac disease, various financial agreements have been reached to fund its progress. Following is a timeline outlining collaborations, licensing agreements and sales related to the drug prior to the recent merger.
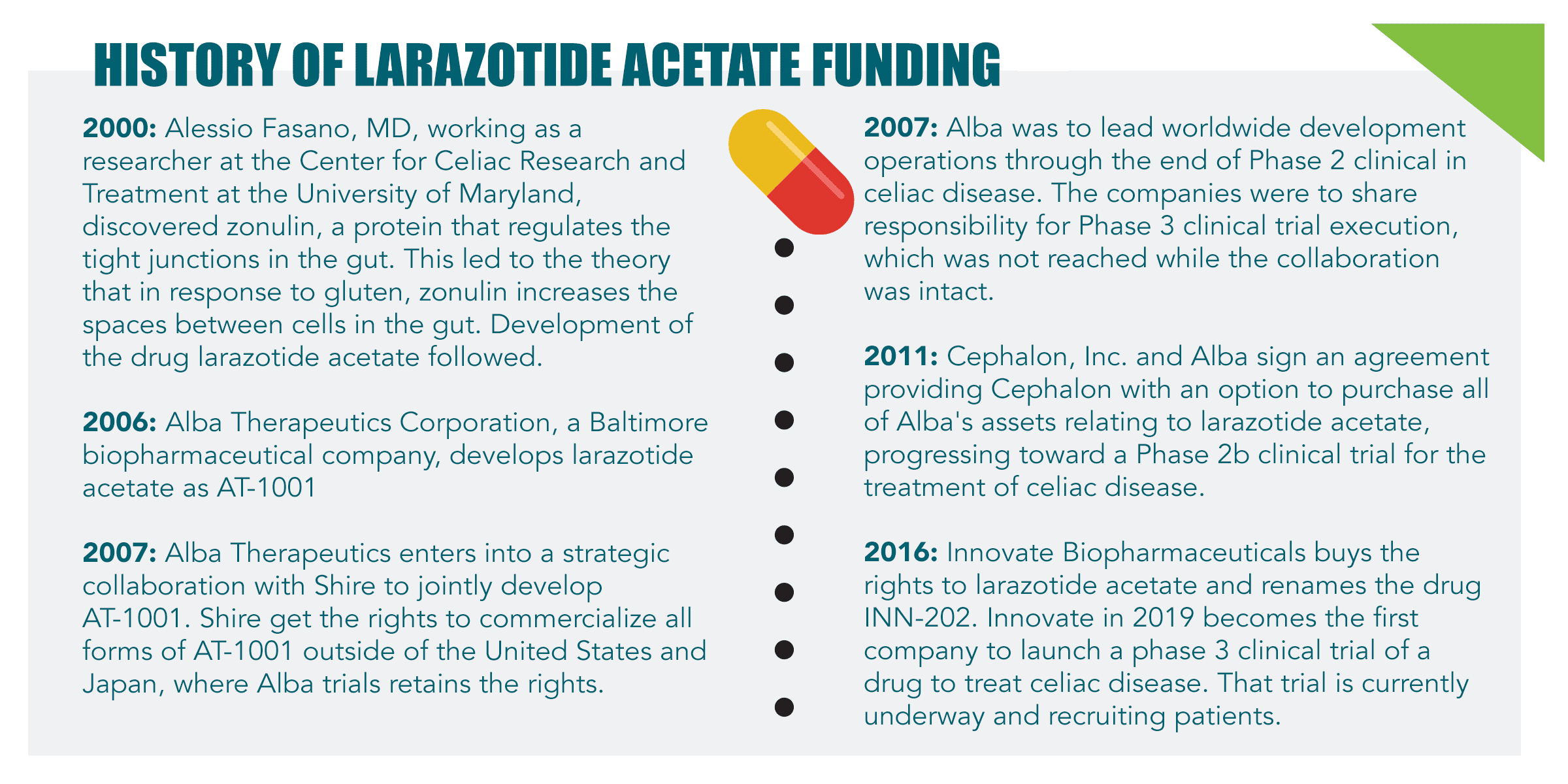
Sitari acquired by GSK
Sitari and GSK have a long relationship since Sitari was the first company formed under a collaboration between GSK and Avalon Ventures. Sitari was founded based on intellectual property from the laboratory of Chaitan Kholsa, Ph.D., professor of chemical engineering, chemistry and biology at Stanford University, and scientific founder of Sitari. Studies for TG2 are in the pre-clinical phase of drug trials.
RELATED: The phases of clinical drug trials |
“Bringing the Sitari TG2 program into GSK is strongly aligned with our research focus on genetically-associated targets and immune-driven disease,” John Lepore, GSK senior vice president of research, said in a press release. “The current standard of care for celiac disease is strict adherence to a gluten-free diet, but a significant number of patients still experience gastrointestinal symptoms and disease progression. TG2 inhibition could represent a new hope for celiac disease patients.”
Muskin noted that multiple strategies are being explored as a treatment that would allow celiac disease patients to ingest gluten without endangering their health. “At Beyond Celiac, we applaud all companies dedicated to this pursuit,” he said. “Beyond Celiac is committed to advancing the understanding of the underlying pathological mechanism of celiac disease through its work with all stakeholders and through its direct support of important research efforts.”