At-Home Test for Celiac Disease Launches
December 05, 2018
Results are designed to be shared with your doctor for a definitive diagnosis

By Amy Ratner, Medical and Science News Analyst
Both Marge Benham-Hutchins and Shannon Myers experienced the all too familiar story of going for years with symptoms of celiac disease before being diagnosed.
Benham-Hutchins’ primary care doctor told her she probably had a nervous stomach. Neither the doctor nor her health insurance company wanted to give her a referral to a specialist, but when she pushed and finally saw a gastroenterologist, he took one look at her symptoms and knew it was likely celiac disease.
Myers’s Vitamin D level was extremely low, she had terrible migraines and worsening vision problems. “I was 50 and I felt 80,” she said. She was called a hypochondriac. Her symptoms were causing her so much anxiety that her primary care doctor wanted her to see a psychiatrist.
In a quest to find out what was wrong, she took a DNA test and found out she was at high risk for celiac disease, something she hadn’t suspected. When she shared the results with her doctor, she was finally tested.
At-home antibody test
A new at-home celiac disease test being launched today by Microdrop Health, a Houston based company, could help patients with similar stories get their doctors to more quickly take the steps needed for diagnosis.
Called imaware™, the test measures the same antibodies to gluten as the tests that doctors use in their offices as the first step to diagnose celiac disease — anti-tissue transglutaminase (tTG) and deaminated gliadin peptide (DGP) tests. In the United States, diagnosis of celiac disease also requires a biopsy that shows intestinal damage.
Some celiac disease experts raised concerns that patients would use the imaware™ test to self-diagnose. But others note that patients, including those who can’t get a doctor to run tests, already often diagnose themselves without any testing after they adopt a gluten-free diet and begin to feel better.
How the test works
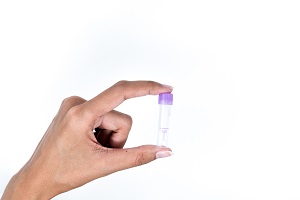 The imaware™ home collection kit, which is sold online only for $99, has two main components, lancets to pierce the skin and a microcontainer to collect a blood sample. Using the lancet, you draw three to four drops of blood from your finger and deposit them in the microcontainer. Only a small sample is needed because the test measures the antibodies in whole blood.
The imaware™ home collection kit, which is sold online only for $99, has two main components, lancets to pierce the skin and a microcontainer to collect a blood sample. Using the lancet, you draw three to four drops of blood from your finger and deposit them in the microcontainer. Only a small sample is needed because the test measures the antibodies in whole blood.
Once the blood is collected, you send it in a pre-paid bag to Microdrop’s CLIA-certified lab. After the sample gets to the lab, it’s processed within 48 hours, with testing done for four celiac disease biomarkers, tTG-IgA, tTG-Igg, DGP-IgA and DGP-IgG.
![]() CLIA-certified lab: A laboratory that is certified by its state and the Center for Medicare and Medicaid Services under the Clinical Laboratory Improvement Amendments (CLIA). Standards are set to ensure the accuracy of results, including qualifications of those who perform the tests. A lab must have CLIA certification before it can accept human samples for diagnostic testing
CLIA-certified lab: A laboratory that is certified by its state and the Center for Medicare and Medicaid Services under the Clinical Laboratory Improvement Amendments (CLIA). Standards are set to ensure the accuracy of results, including qualifications of those who perform the tests. A lab must have CLIA certification before it can accept human samples for diagnostic testing
,
including qualifications for individuals who perform the test and other standards that ensure the accuracy and reliability of results.
Quantitative results from the celiac disease tests used in imaware™ are aggregated and likelihood ratios are applied to determine the probability that you have celiac disease. Several studies have shown that the use of likelihood ratios improves the clinical interpretation of blood tests for celiac disease and diagnosis.
![]() Likelihood ratio: A ratio of the probability that a test result is correct to the probability that the test result is incorrect. The sensitivity and specificity of the test are used to generate a likelihood ratio, which is calculated for both positive and negative test results. Likelihood ratios are used to assess the potential utility of a diagnostic test and how likely it is that a patient has a disease or condition.
Likelihood ratio: A ratio of the probability that a test result is correct to the probability that the test result is incorrect. The sensitivity and specificity of the test are used to generate a likelihood ratio, which is calculated for both positive and negative test results. Likelihood ratios are used to assess the potential utility of a diagnostic test and how likely it is that a patient has a disease or condition.
You have to be consuming at least 2 grams of gluten per day – the equivalent of 1.5 slices of regular bread – for at least eight weeks in order for the tests to be accurate, the company says. When ordering the test, you are asked to verify that you are not on a gluten-free diet. You or a guardian can also provide information about other risk factors for celiac disease, including having: a first degree relative who has been diagnosed; type 1 diabetes; or Down syndrome.
When lab results are ready, you get an email that links to a password-authenticated online portal. Once you’ve signed in, you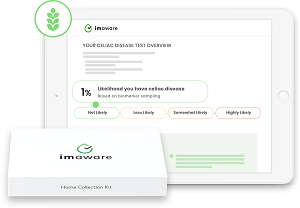 can access a report that tells you whether you what the chance is that you have celiac disease. For example, it may say you are highly likely or unlikely to have celiac disease.
can access a report that tells you whether you what the chance is that you have celiac disease. For example, it may say you are highly likely or unlikely to have celiac disease.
The report also details the likelihood ratio in four categories: highly likely with a ratio of greater than 60 percent; somewhat likely, 20 to 60 percent; less likely, 2 to 20 percent and unlikely, 0 to 2 percent.
The report also includes the quantitative and qualitative – positive or negative – results for each of the tests run.
The test results are not definitive for a diagnosis of celiac disease and consumers are advised to share the results with their doctor for follow-up.
“The test is not intended to diagnose celiac disease – only your healthcare provider can make that determination in light of your overall health history and the results of other tests they may decide to order,” the company says in the test result report.
Microdrop also plans to eventually offer the test to those already diagnosed with celiac disease as a way to monitor compliance with the gluten-free diet by detecting antibodies that indicate exposure to wheat, barley and rye.
The imaware™ test is considered a laboratory developed test, which the U.S. Food and Drug Administration defines as a diagnostic test that is designed, manufactured and used within a single laboratory. Tests in this category aren’t subject to FDA pre-market review or other regulations because they traditionally have been relatively simple lab tests available on a limited basis. But with advances in technology tests related to medical conditions are becoming more complex and widespread, and since 2010 the FDA has been reviewing its policy regarding oversight, though no action has been taken.
Accuracy of the test

Sensitivity and specificity are the two standards by which accuracy of testing is measured. Overall, imaware™ reports sensitivity of 95 percent and specificity of 99 percent.
![]() Sensitivity – The ability of a test to correctly classify a person as having a disease
Sensitivity – The ability of a test to correctly classify a person as having a disease
Specificity – The ability of a test to correctly classify a person as not having a disease
The imaware™ test has been through review in four studies, two internal and two by independent third parties, according to the company. Nearly 1800 samples have been tested, including those provided by consumers participating in a pre-launch “friends and family” review of the test, and those collected from Massachusetts General Hospital for Children and the Celiac Center at Beth Israel Deaconess Medical Center. Study results have been submitted to a peer reviewed medical journal for publication consideration.
Blood samples collected from biopsy-confirmed celiac disease patients were run through imaware™ as part of the test validation process, said Jani Tuomi, chief digital officer and company spokesman.
“The test performance appears to be very good compared to the established clinical lab-based tests used in the United States and Europe,” said Detleff Schuppan, M.D., director of the Institute of Translational Immunology and Celiac and Small Intestinal Diseases at the University of Mainz in Germany and a medical advisor to Microdrop. “There is no real difference.”
He said that if someone in the general population has a positive tTG result, the probability that the person has celiac disease is 95 percent to 99 percent. If the test is used by someone already diagnosed with celiac disease to determine whether gluten is getting into their gluten-free diet, the sensitivity is 99 percent or greater, according to Schuppan.
Stephano Guandalini, M.D., founder of the University of Chicago Celiac Disease Center and also a Microdrop medical advisor, said the reliability of the test stems from its use of the tTG IgA antibody, which is universally recommended for diagnosis of celiac disease and has “unsurpassed sensitivity.”
Improving the diagnosis rate
Despite some gains in recent years, celiac disease has long been plagued by low diagnosis rates, with estimates that less than 20 percent of patients who have the condition know it. Patients often report that their doctors do not want to test them despite symptoms. Even when a family member has celiac disease, a known risk factor for the genetic, autoimmune condition, physicians can be reluctant to test. The average delay in diagnosis is six to 10 years.
A study published recently in the United European Gastroenterology Journal concluded that patients whose diagnosis had been delayed for three years reported being sick more often, going to the doctor more frequently and using more drugs for dyspepsia and depression than those who were diagnosed more quickly.
When Microdrop was developing plans for use of its testing technology, the company was specifically interested in under-diagnosed diseases where it felt it could make a significant impact, Tuomi said. The celiac disease test is the first being launched, with separate tests for rheumatoid arthritis, prostate cancer and heart disease to follow.
“We said, ‘Let’s go somewhere where we think we can really help out,’” Tuomi said. “We did not wake up one day saying celiac disease would be our first test.”
The company turned to the scientific community, according to Tuomi, and created a scientific advisory board made up of celiac disease experts Schuppan and Guandalini.
“I do believe that a more widespread testing for celiac disease is a good thing. In this country we still lag behind in our diagnostic rates,” Guandalini said, putting the diagnosis rate at less than 50 percent. “A reliable screening test freely available to consumers seems to me as step in the direction of improving diagnosis rates.”
“The possibility of reliable home testing as an initial screen for celiac disease could help thousands of individuals to understand whether they are suffering from this condition,” said Marie Robert, M.D., Beyond Celiac chief scientific officer.
Physician follow-up needed
Benjamin Lebwohl, M.D., director of clinical research at the Celiac Disease Center at Columbia University, who is not connected to Microdrop, agreed that the technology might lead to an improvement in diagnosis rates. “We don’t yet know how these tests will be used, but I believe they could prove to be useful,” he said.
In particular, the at-home test could help patients who have symptoms and a family history of celiac disease, but limited access to a physician who is knowledgeable about celiac disease, according to Lebwohl. “A positive result could prompt the person to find [a doctor experienced in celiac disease] and initiate full testing,” he said.
But he noted it is too early to tell how truly reliable the home tests are. And he raised concern that someone who gets a positive result could overestimate its accuracy, assume he or she had celiac disease and skip further testing and the biopsy required for celiac disease diagnosis.
“This could lead to a needless, long-term gluten-free diet in false positive patients,” he said, adding that false positives could also lead to needless biopsies and worry for patients.
Ed Liu, M.D., director of the Colorado Center for Celiac Disease, shares Lebwohl’s concerns. He said the potential that patients would assume they are definitively diagnosed or cleared of having celiac disease by the test is “a major problem.”
“This will further the notion that this is a condition that is entirely managed by patients on their own with the diet, without any medical monitoring or supervision,” he said.
Guandalini agreed there is a risk that patients could over-interpret the screening test as a diagnostic test, but he said the results report clearly says that it “cannot and should not be interpreted as a definitive diagnosis.”
On the other hand, the test could help reduce the number of people who put themselves on the gluten-free diet without diagnosis, Guandalini noted.
“One of the reasons people may choose to begin a gluten-free diet is that either it takes too long to be seen by a gastroenterologist, or that the healthcare provider may not be inclined to check the potential patient for celiac disease,” he said. “In these circumstances, having the chance to be immediately and easily tested may be a useful tool to prevent a self-prescribed gluten-free diet.”
Both he and Schuppan emphasized that the test report advises patients to follow-up and share results with a physician knowledgeable about celiac disease.
“It is extremely important. At-home tests can only be considered a first step in the diagnostic process for celiac disease, never a conclusive one,” Guandalini said, adding that follow-up should take place whether results are positive or negative.
“The most important aspect of using such a test is to discuss the results with a professional health care provider (physician, nurse practitioner or physician assistant) with knowledge of celiac disease,” Robert said.” Self-diagnosis is to be avoided.”
It’s unclear whether a physician would re-do the celiac disease blood tests. Schuppan said he thinks it would be unnecessary based on the accuracy of the results, but the test results report notes only your doctor can make a diagnosis based on your overall health and other tests that may be ordered. The report also advises patients not to make any dietary changes before consulting a healthcare provider.
In addition to the biopsy necessary for definitive diagnosis, celiac disease patients should be evaluated for other autoimmune related conditions and nutritional deficiencies, additional reasons follow-up is so important, according to Schuppan. But he noted that these steps are not always taken even when patients get initial blood tests from doctors in their offices.
Liu worried that if the test shows a low likelihood of celiac disease, lack of follow up could cause a missed diagnosis of celiac disease and any related condition. “How often would follow-up advice be heeded?” he asked.
“If it’s a reliable test that gives you clear results for celiac disease, why not [take it]?” Schuppan asked. “I think it makes a lot of sense.”
Microdrop is a corporate sponsor of the Society for the Study of Celiac Disease (SSCD) and will work with the group to promote the use and awareness of scientifically validated approaches and methods for celiac disease diagnosis, according to the SSCD website. The SSCD is an organization of medical, scientific and allied health professionals in the field of celiac disease.
Barbara Connell, chief operating officer, said the organization is not currently conducting any studies using the test. Nor does the SSCD endorse the use of the test or any other product or have any statement on the validity of the test, Connell said.
“Beyond Celiac is partnering with imaware™ to support the ambitious goal of dramatically reducing the number of individuals dealing with undiagnosed celiac disease by helping them more quickly take the steps needed for diagnosis,” said Alice Bast, Beyond Celiac chief executive officer. The company will contribute a portion of sales to Beyond Celiac to support ongoing initiatives to drive diagnosis, advance research and accelerate the discovery of new treatment and a cure.
Other at-home tests for celiac disease
While Microdrop is the first U.S. company to offer direct-to-consumer blood testing for antibodies to celiac disease, PrivaPath Diagnostics, a company headquartered in Ireland, does antibody testing through its Let’s Get Checked product. The company offers a variety of tests in addition to celiac disease.
Other celiac disease tests on the market use saliva samples to determine if you have genes that usually found in people who have celiac disease. 23and Me and other DNA tests for celiac disease are designed to detect the HLA-DQ2 and HLA-DQ8 genes. About 95 percent of people with celiac disease have the HLA-DQ2 gene and most of the remaining 5 percent have the HLA-DQ8 gene.
However, the genes are very common even in those who don’t have celiac disease — about 40 percent of the population has the genes, but only about 5 percent of those with the genes have celiac disease. A positive genetic test for celiac disease requires follow-up with antibody testing and biopsy for diagnosis.
Unlike antibody blood testing, you do not need to be consuming gluten for DNA testing to be accurate.
In 2017, the FDA approved 23andMe’s genetic test for celiac disease and nine other diseases and conditions. The approval, which cleared the way for the company to market tests to assess genetic risks, came after the FDA conducted a review of studies showing consistent link between certain gene variants and the 10 diseases and conditions. The FDA also reviewed data to show that the test was accurate and provided reproducible results. This criterion could eventually also be applied to laboratory developed tests like imaware™, something Tuomi said Microdrop is preparing to provide.
Patient perspectives
For Shannon Myers, a DNA test turned out to be on-target. After getting her results from 23andMe, she took them to her doctor and was tested for and diagnosed with celiac disease. Her experience leads her to think at-home testing for celiac disease patients can provide the impetus or evidence for diagnosis.
Although she is a nurse practitioner, Myers said she had not considered celiac disease as the possible cause of her symptoms. When she was in training, celiac disease was still described as a rare condition that mainly affected children who had failure to thrive. Her DNA result was a complete surprise.
Consequently, she might not have thought to take a test like imaware™even if had existed. She thinks the test might have the most appeal to people who have family members diagnosed with celiac disease but still can’t get their doctor to test them. A member of the Beyond Celiac Patient and Family Advisory Council, she’s heard this story often, including from her brother whose doctor did not want to test him because he did not have diarrhea.
Myers she said she could envision a scenario in which someone who had many symptoms and got back clearly positive results would not follow up with a physician in order to protect themselves from having a pre-existing condition that might cause insurance problems. “Maybe in certain cases I would try the test and keep the [positive] test results to myself,” she said.
Marge Benham-Hutchins, an academic researcher who is also on the advisory council, said she had negative blood tests only a few days after a biopsy that showed total destruction of the absorbing villi in her intestine. A test like imware™ would not have been helpful to her, she said, noting her situation is not typical. But she is interested in learning more about the test and the company that makes it.
The test could be valuable for some people, and it might wake up healthcare providers in regard to diagnosis of celiac disease, she said.
“I am all for people having access to medical tests and making intelligent decisions without having to filter everything through the doctor,” she said. “And if you get the results and go on the gluten-free diet, at least that’s better than not knowing at all that you have celiac disease.”


