
Note from Alice: Much to Be Thankful For

November is a great month for reflection. The end of the year is in sight and Thanksgiving is just around the corner. This year, the entire Beyond Celiac team, Board and I have so much to be thankful for, and my November column is the perfect place to share from our long list of gratitude.
First, we are so thankful for all the volunteers, runners, vendors and sponsors that made our Step Beyond Celiac 5Ks a wonderful success this year. This was our third year in Kansas City, an amazing race that was created from nothing by then-volunteer Jilian Hunt and our board member, Chad Shaffer, MD, and built into an annual not-to-miss event. It was also our second Virtual 5K, and our first year in Philadelphia and Dallas. All our races went off without a hitch and raised thousands of dollars for celiac disease research. We are extremely grateful for all the hard work everyone put in and can’t wait to see how these events continue to grow, expand and flourish in 2020! If you would like to volunteer, we’d love to have your participation.
We are also thankful for our ambassador, Arturo Chacon-Cruz. Despite being an incredibly busy global opera star, Arturo has dedicated a great deal of time to championing Beyond Celiac and our mission throughout this past year. He has been a wonderful partner in raising celiac disease awareness and helping to raise funds for our research grants program. Most notably, he recently performed beautifully at our Go Beyond Celiac Gala & Concert. The benefit was an amazing night, one that we will never forget! The day following the concert, calls and emails came pouring in praising us for such a special evening.
Additionally, we are grateful to all the researchers working in their labs tirelessly to try to unlock the mysteries of this serious autoimmune disease. We very much appreciate the work of the scientists striving for treatments and a cure beyond the gluten-free diet. The field of celiac disease research is woefully underfunded (one more reason we started our Research Grants program) and we want to recognize the efforts of those who continue on despite the many challenges and obstacles in their paths. We are thankful and hoping for a brighter future where we can live our lives to the fullest and eat without fear.
Lastly, we are so thankful for YOU. Our community is what keeps us going. We are so grateful for all of our donors, our grassroots fundraisers, our monthly contributors, our social champions, our email readers, our event participants, our advisory boards and our many volunteers. You make it possible for us to keep going. As a nonprofit, Beyond Celiac and our programs and research grants are supported by people like you. We recognize how much our community has done for us this year, making it possible for us to fund research, hold the 2019 Symposium and Summit, create and launch the Go Beyond Celiac app, expand our 5Ks, and offer free and up-to-date materials and web content with the latest info and power our programs. We are so thankful that we are in this together for a cure.
Thank you all!
To gratitude and living life Beyond Celiac,
Alice Bast

From the Chief Scientific Officer (CSO), Marie Robert, MD

One of our most important roles at Beyond Celiac is making sure doctors, researchers, pharmaceutical companies, and other key stakeholders listen to patients and understand the needs of the celiac disease community. At our May 2019 Research Symposium, Linda Deal, MS, of Pfizer discussed the importance of including patients as research partners. Patient engagement in the drug development process ensures any treatment in the drug development pipeline will meet the needs of people with celiac disease. It also leads to clinical trials that measure outcomes important to patients.
At our upcoming Research Summit, we invited people with celiac disease to speak directly to doctors, researchers, nutritionists, pharmaceutical companies, insurance companies, and the FDA. Our Summit, titled Getting Over the Peak: The Final Ascent to Realizing Therapies in Celiac Disease, will discuss the remaining barriers to bringing a celiac disease treatment to market and how we can work together with all stakeholders to overcome these barriers. On the heels of new study results from Cour, and one potential treatment in the furthest-ever stage of drug development, this is a very timely topic.
On the morning of November 15, we will be live webcasting two vibrant patient panel discussions at the Summit. In the first panel, we will hear from people with celiac disease and from parents of children with celiac disease. Moderated by Anne Lee, EdD, RDN, LD, the panelists will discuss their celiac disease experiences, including how they navigate the gluten-free diet, how they eat out, and how their lives have changed since they or their children were diagnosed.
The second patient panel will feature people who have participated in celiac disease clinical trials. Our four panelists will discuss how they learned about the clinical trial, what their participation entailed, and if they would participate again. We’ve heard before from researchers about the need for patient participation to make clinical trials successful. If you’ve ever wondered what it would be like to be part of a clinical trial, you’ll definitely want to tune in.
In between the two panels Joseph Murray, MD, of the Mayo Clinic and international celiac disease expert will discuss the current status of celiac disease drug development, including an overview of the barriers to progress.
You can sign up for the webcast here. If you sign up, you will receive a link that will let you watch live or watch the recording afterward.
The Summit will also feature discussions of the often-ignored question of how and when to include children in clinical trials and the role that patient advocacy groups play in advancing research internationally.
Other highlights will include representatives from the Food and Drug Administration discussing the role of the patient voice in drug development. We will also hear from pharmaceutical companies that have potential treatments in the drug development pipeline. The Summit will be interactive, with lots of time for questions and comments from the entire in-person audience, further making sure the celiac disease community members in attendance have a chance to have their views heard.
Our goal is to leave the Summit with specific immediate, short term and long term actions that the entire celiac disease community will tackle, both individually and together to overcome the remaining barriers to bringing a treatment beyond the gluten-free diet to the celiac disease community. A detailed report of the meeting, explicitly stating both the barriers to therapies and the strategies to overcome those barriers will be posted on our website.
Thank you to our Summit Gold Sponsor, Takeda.
 Chef Oonagh Williams holds a culinary arts degree and spends her time cooking and educating the public on gluten- and allergy-free diets. She herself has celiac disease and food allergies. She often teaches cooking classes, hosts dinner parties, and offers one-on-one help. Buy her Delicious Gluten-Free Cooking e-book and connect with her on Facebook.
Chef Oonagh Williams holds a culinary arts degree and spends her time cooking and educating the public on gluten- and allergy-free diets. She herself has celiac disease and food allergies. She often teaches cooking classes, hosts dinner parties, and offers one-on-one help. Buy her Delicious Gluten-Free Cooking e-book and connect with her on Facebook.
Research News
Researchers from the Celiac Disease Program at Children’s National Health System and the Harvard Celiac Disease Program had celiac disease patients like these in mind when they did a recent study that investigated whether a separate toaster and other kitchen items are needed.
Raise Money for Beyond Celiac on Facebook!
 Happy birth-month to all our November-born community members! When your big day rolls around, consider “donating” it to Beyond Celiac so that we can help fund new research scientists in the celiac disease field. And a HUGE thank you to everyone who has created Facebook fundraisers for us this year!
Happy birth-month to all our November-born community members! When your big day rolls around, consider “donating” it to Beyond Celiac so that we can help fund new research scientists in the celiac disease field. And a HUGE thank you to everyone who has created Facebook fundraisers for us this year!
Overcoming Motherhood Imposter Syndrome
 Actress Casey Wilson shares her son’s harrowing celiac disease diagnosis journey with New York Times Parenting.
Actress Casey Wilson shares her son’s harrowing celiac disease diagnosis journey with New York Times Parenting.






 A tasty fall treat that makes a fun addition to your Thanksgiving dessert table!
A tasty fall treat that makes a fun addition to your Thanksgiving dessert table! A warm, autumn-themed dessert to keep you feeling cozy as the nights start to turn cold!
A warm, autumn-themed dessert to keep you feeling cozy as the nights start to turn cold! Chef Oonagh Williams holds a culinary arts degree and spends her time cooking and educating the public on gluten- and allergy-free diets. She herself has celiac disease and food allergies. She often teaches cooking classes, hosts dinner parties, and offers one-on-one help. Buy her
Chef Oonagh Williams holds a culinary arts degree and spends her time cooking and educating the public on gluten- and allergy-free diets. She herself has celiac disease and food allergies. She often teaches cooking classes, hosts dinner parties, and offers one-on-one help. Buy her
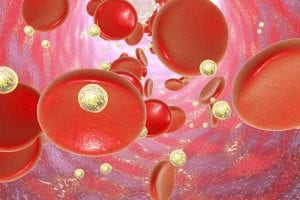 A potential treatment for celiac disease in which nanoparticles act like a Trojan horse and carry a hidden component of gluten to reprogram the immune system not to react has shown positive early results in a new clinical trial.
A potential treatment for celiac disease in which nanoparticles act like a Trojan horse and carry a hidden component of gluten to reprogram the immune system not to react has shown positive early results in a new clinical trial.

 Three flavors of Simply Asia Street Noodles that have the Beyond Celiac gluten-free trademark are being voluntarily withdrawn from the market by the manufacturer because they contain an ingredient not allowed under the trademark rules.
Three flavors of Simply Asia Street Noodles that have the Beyond Celiac gluten-free trademark are being voluntarily withdrawn from the market by the manufacturer because they contain an ingredient not allowed under the trademark rules. On Wednesday, November 13th, 2019 the Digestive Disease National Coalition (DDNC) will coordinate a National Day of Advocacy to encourage federal legislators to address step therapy reform by supporting the Safe Step Act.
On Wednesday, November 13th, 2019 the Digestive Disease National Coalition (DDNC) will coordinate a National Day of Advocacy to encourage federal legislators to address step therapy reform by supporting the Safe Step Act. Happy birth-month to all our November-born community members! When your big day rolls around, consider “donating” it to Beyond Celiac so that we can help fund new research scientists in the celiac disease field. And a HUGE thank you to everyone who has created Facebook fundraisers for us this year!
Happy birth-month to all our November-born community members! When your big day rolls around, consider “donating” it to Beyond Celiac so that we can help fund new research scientists in the celiac disease field. And a HUGE thank you to everyone who has created Facebook fundraisers for us this year! Actress Casey Wilson shares her son’s harrowing celiac disease diagnosis journey with New York Times Parenting.
Actress Casey Wilson shares her son’s harrowing celiac disease diagnosis journey with New York Times Parenting.

